 
For future date, specify interval value 2 hours from the current timestamp: now() + interval ‘2 hour’.For example, the below SQL query gives the following values. You can also specify an interval for returning the date after a specified period. Now()-Orderdate as Interval FROM SalesOrders For example, the query below returns the interval between the current timestamp and from the SalesOrders table. You can use the minus (-) operator to calculate the difference between two dates. SELECT TO_CHAR(NOW() :: DATE, 'dd-mm-yyyy') You can combine TO_CHAR() and the Now() function to convert the current timestamp into the specified format. You can specify double colons (::) to cast a DATETIME value to a DATE value. The Now() function returns the current system timestamp (date and time). To_char(OrderDate,'DDD MM/DD/YYYY') As "DDD MM/DD/YYYY" To_char(OrderDate,'DAY MM/DD/YYYY') As "MM/DD/YYYY", To_char(OrderDate,'MM/DD/YYYY') As "MM/DD/YYYY", To_char(OrderDate,'DD.MM.YY') As "DD.MM.YY", To_char(OrderDate,'DD-MM-YYYY') As DDMMYYYY, SELECT Orderdetails,OrderDate as ExistingDateformat, Month abbreviation in all capital, first letter capitalized and all lowercase letters, respectivelyĭay name in all capital, first letter capitalized and all lowercase letters, respectivelyĭay of the week (Sunday (1) to Saturday (7))Ī few examples of different date formats are specified in the following SQL. 
Month name with the first letter capitalized The supported values in the to_char() function are as shown below. To_char(OrderDate,'DD-MM-YYYY') As NewDateFormat FROM SalesOrders  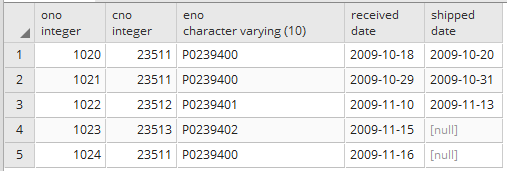
The following query converts existing date values stored in the table into DD-MM-YYYY format.
0 Comments
 
Once the download completes, the installation will start and you'll get a notification after the installation is finished. BATTLE BEARS -1 will be downloaded onto your device, displaying a progress.A pop-up window with the permissions required by BATTLE BEARS -1 will be shown.Tap on the Install button located below the search bar and to the right of the app icon. Once the BATTLE BEARS -1 is shown in the Google Play listing of your Android device, you can start its download and installation.Use the indicated client to activate key and download and play your game. Head over to one of the trusted game stores from our price comparison and buy cd key at the best price. Click on the Continue To App button on our website. Compare prices with GG.deals to find the cheapest cd key for Battle Bears 1: Zombies PC.How to install BATTLE BEARS -1 on your Android device: Your antivirus may detect the BATTLE BEARS -1 as malware as malware if the download link to 1 is broken. We have already checked if the download link is safe, however for your own protection we recommend that you scan the downloaded app with your antivirus. Download the Game Channel app to get a FREE game every day SkyVu presents BATTLE BEARS ROYALE, the first class based team multiplayer shooter for iPhone. The app is listed on our website since and was downloaded 453 times. To install BATTLE BEARS -1 on your Android device, just click the green Continue To App button above to start the installation process. This app was rated by 5 users of our site and has an average rating of 4.8. The latest version released by its developer is 1.4.1. The company that develops BATTLE BEARS -1 is SkyVu Inc. Adjust SENSITIVITY SLIDER in Options if needed.īATTLE BEARS -1 is a free app for Android published in the Arcade list of apps, part of Games & Entertainment. To Fire: Use RIGHT Thumb to HOLD DOWN the Red Joystick. To Look Around: Slide RIGHT Thumb anywhere outside of Red Joystick. HOW TO PLAY - STYLE 1: To Move: Slide LEFT Thumb over Green Joystick. "Every action fan will want to grab a copy of BATTLE BEARS -1 for its hilarious style and non-stop rainbow-spewing" -AppSpy "The guys and girls at SkyVu have a spooky ability to create the most compulsive and playable games available." "BATTLE BEARS -1 is a game that you should NOT pass up on!" -TouchAholics Defend the flagship Ursa Major from an onslaught of confused creatures hewn from the deepest, darkest corners of the cosmos' disturbed imagination.īATTLE BEARS -1 is an action-packed 3D shooter with massive bosses, heaps of humor, tons of crazy weapons, and no shortage of fluffiness or shooting rainbows. Play with 8 players & stake your claim as the best BATTLE BEAR in the world OVER 25 MILLION DOWNLOADS 'A funny action shooter everyone can play.' 'A Complete Multiplayer Experience' -AppPicker 'A cross between COD & TMNT.'-Wall Street Journal. Join OLIVER, RIGGS and WIL on a space odyssey none of them are anywhere close to thrilled to be experiencing. Just enjoy Battle Bears Royale PC on the large screen for free Battle Bears Royale Introduction. Get BATTLE BEARS Negative One! The most intense 3D shooter for your high-end Android device! The #1 Action/Adventure Game in the USA, UK, Canada, Australia, and China! Nominated for the BEST APP EVER AWARD.   
Peaks can therefore be assigned to specific bonds, allowing identification of an unknown compound from the IR spectrum. However, each type of bond will absorb IR radiation within a specific frequency range, and will have a common peak shape and absorption strength. The absorbed frequencies depend on the identity and electronic environment of the bonds, giving each molecule a characteristic spectrum. In this type of spectrum, the peaks are inverted, as they represent a decrease in transmitted light at that frequency. This is generally represented with a spectrum of percent IR radiation transmitted through the sample at a given frequency in wavenumbers. An IR spectrophotometer can measure which frequencies are absorbed. Molecules with certain types of covalent bonds can absorb IR radiation, causing the bonds to vibrate. Infrared, or IR, spectroscopy is a technique used to characterize covalent bonds. Characteristic IR frequencies of covalent bonds present in organic molecules.įigure 2. Diagram showing C - O double and C - C triple bond stretches and the resulting change in the dipole moment. This method enables one to directly examine solid or liquid analytes without further preparation.įigure 1. Typically, materials with a high refractive index are used, such as germanium and zinc selenide. In this experiment, the ATR sampling technique is used, where the infrared light reflects off the sample that is in contact with an ATR crystal multiple times. Notice the peak at 3,343 cm -1 for the N-H single bond and the peak at 1,695 cm -1 for the carbonyl groups. Figure 2 shows the IR spectrum of a Hantzsch ester. Table 1 shows some characteristic absorption frequencies. Hence, a carbonyl group stretch will show an intense band in the IR, and a symmetrical internal alkyne will show a small, if not invisible, band for stretching of the C-C triple bond ( Figure 1). The intensity of the infrared absorption is proportional to the change in the dipole moment when the bond stretches or compresses. On the other hand, a symmetrical alkyne does not have a net dipole moment because the two individual dipole moments on each side cancel each other.  
Hence, there is a net dipole moment resulting in a partial negative charge on oxygen and a partial positive charge on carbon. For example, in a C-O double bond (a carbonyl group), the electrons spend more time around the oxygen atom than the carbon atom because oxygen is more electronegative than carbon. If the electronegativities (the tendency to attract electrons) of the two atoms in a covalent bond are very different, a charge separation occurs that results in a dipole moment. When a molecule absorbs infrared light with a frequency that equals the natural vibrational frequency of a covalent bond, the energy from the radiation produces an increase in the amplitude of the bond vibration. Infrared light is electromagnetic radiation with wavelengths ranging from 700 nm to 1 mm, which is consistent with the relative bond strengths. Double and triple bonds can be considered as stronger springs, so a C-O double bond has a higher stretching frequency than a C-O single bond. Thus, C-H, N-H, and O-H bonds have higher stretching frequencies than C-C and C-O bonds, as hydrogen is a light atom. The frequency is typically measured in wavenumbers, which are expressed in inverse centimeters (cm -1).įrom Equation 1, the frequency is proportional to the strength of the spring and inversely proportional to the masses of the objects. This frequency is given by Equation 1, where k is the force constant of the spring, c is the speed of light, and µ is the reduced mass ( Equation 2). Naturally, this bond stretches and compresses with a certain vibrational frequency. The following table lists infrared spectroscopy absorptions by frequency regions.A covalent bond between two atoms can be thought of as two objects with masses m 1 and m 2 that are connected with a spring.   
But they have a lot of downsides for inside the waistband carry. I should clarify the FN 509 Tactical open-ended holsters are fine for OWB carry. But an open-ended holster is going to have a couple of issues.įor one, if you’re wearing the holster inside the waistband, it’s not going to be as comfortable. If you have an open-ended holster, it isn’t a big deal as far as compatibility. Now the FN 509 Tactical is a different animal from these other guns due to the threaded barrel. You can use pretty much any FN 509 holster with this gun. Again, FN did a great job making sure your holster compatibility wouldn’t be an issue. It has a compact-length grip that holds 15 rounds instead of 17. The Mid-size gun is a full-length four-inch slide like the FN 509 Tactical. The only difference between the Compact and the Midsize are the optics cut. The FN 509 Midsize is essentially the same gun as the FN 509 Compact MRD. So any 509 holsters with an optics cut will work for this firearm. So you won’t have to look for a shorter slide over other guns. The slide itself is a standard FN 509 slide. So you want to make sure your holster is optics compatible. So if you’re going to run the MRD, you’re likely going to be running an optics. And what’s unique about the MRD holster is that it has an optics cut. Each high-performance feature was critically chosen to give shooters every advantage.The slide on the FN 509 Compact is the same size as any other gun. The pistol features the newest Long Slide from FN and this feature makes the FN 509 LS Edge the most advanced striker fired 509 handgun available. The slide features lightening cuts to reduce weight and improve balance when it comes to consecutive shots. The FN 509 LS Edge boasts enhanced ergonomics and adaptability, which means it’s designed to fit any shooter. The pistol comes with a double stack magazine, which can be loaded with up to 17 rounds of the 9x19mm Parabellum cartridge. Its overall length is 8.2 inches and its barrel is 5 inches long. The FN 509 LS Edge is a full-sized handgun. The pistol features an optimized long slide with patented optics mounting system, which makes it a great choice for fans of red dot, RMR, and reflex sights. The FN 509 LS Edge is one of the best tactical pistols currently available in the market. The pistol was introduced only a couple of months ago (in 2021) and it belongs to the extremely popular FN 509 family of pistols. The FN 509 LS Edge is a double-action, striker-fired, semi automatic pistol developed by FN Herstal. Browse our whole selection of FN.īrowse our whole selection of FN Holsters. Our FN 509 LS Edge - 5" holsters are available in many varieties, and carry styles covering everything a gun owner needs, including IWB, OWB, Cross Draw, Shoulder, Small Of Back, Light Bearing, Red Dot Sight, Chest, Drop Leg, Duty, Light & Red Dot, Paddle, Other, Concealed Carry, Ankle, Appendix, Competition, Clip On, Hybrid, Magazine, Minimalist, Open Carry, Sweat Guard, Thumb Break, Tuckable, Women's holster designs.įN 509 LS Edge - 5" OWB holsters are custom made for this gun model for a perfect fit, smooth draw and re-holstering and allows you to carry your gun in comfortable and safe way. 
Strong side holsters, so coalled thumb break holsters, are a fan-favorite among shooters around the world as placing your FN 509 LS Edge - 5" on your hip, in a high ride holster, pancake holster or a side holster is almost natural and this style of carry fits your everyday carry needs. 
509 LS Edge - 5" belt holsters are a classic and proven way of carry. The path to chose the right FN 509 LS Edge - 5" OWB holster is not so easy. Other than that, we have a large range of on-duty or off-duty holsters for your daily carry. Our FN 509 LS Edge - 5" holsters are designed to minimise gun print so your carry is only known to you, their slim profile and premium stitching are a key feature. Wide range of carry positions and carry styles allow you to browse through many holster models with great retention and a covered trigger guard, either open top holsters or holsters with a thumb break. Our offer includes 103 designs of FN 509 LS Edge - 5" holsters with key features such as quick and easy draw and re-holstering.   Synchronization isn't available as far as I know you'll need to manually import any new classes. One very important setting is Arrange when you drop the classes they get a cramped right click on any empty space at a class diagram and click Arrange AllĪrrows in the model explorer view can be grabbed to the diagram to show generalization, realization etcĪfter all of that your settings will show diagrams like You can have default friendly color settings from Window -> pereferences -> papyrus -> class diagram To show the class elements (variables, functions etc) Right click on any class -> Filters -> show/hide contents Voila !! Then start grabbing classes to the diagram from the view In the view Right click root model -> New diagram In that view, you'll find your classes like in the picture What we did in here was that we replaced an empty model with a generated one.ĪLT+W -> show view -> papyrus -> model explorer 
Move generated MyProject.uml which was generated at root, to UML folder, Eclipse will ask you If you wanted to replace it click yes. Right click on MyProject_kdm.xmi -> Discovery -> Discoverer -> Discover UML model from KDM code again you'll get a property dialog set the serialization prop to TRUE to generate a file named MyProject.uml Move the three generated files MyProject.di, MyProject.notation, MyProject.uml to the UML folder.Right click project name file -> new -> papyrus model -> and call it MyProject.Right click over the project name -> Discovery -> Discoverer -> Discover Java and inventory model from java project, a file called MyProject_kdm.xmi will be generated.In your java project (assume it's called MyProject) create a folder e.g UML.Repeat steps 1-3 and this time, install Modisco.After installation finishes restart Eclipse.In the Work with: drop-down, select -All Available Sites.I wish papyrus had straightforward way to Reverse Engineer classes into UML class diagram and It would be super cool if that reverse engineering had a synchronization feature, but unfortunately papyrus project is full of features and I think developers there have already much at hand since also many actions you do over papyrus might not give you any response and just nothing happens but that's out of this question scope anyway. UML generation (with synchronization feature) is really important not to software designer but to the average developer. Since I'm going back to do some modeling and also getting some replies to the post I decided to install papyrus again and will investigate other possible UML modeling solutions again. 
My original post was in June 2012 a lot of things have changed many tools has grown and others didn't. If you're a designer then Papyrus is your best choice it's very advanced and full of features, but if you just want to sketch out some UML diagrams and easy installation then ObjectAid is pretty cool and it doesn't require any plugins I just installed it over Eclipse-Java EE and works great !. Information is shown in other views wherever applicable. You can drag Java classes from any other view onto the diagram, and diagram-related Diagrams are fully integrated into the Eclipse IDE.If necessary, they are checked out of your All diagrams in your Eclipse workspace are updated with refactoring changes as appropriate.When you rename a field or move a class, your diagram simply reflects the Refactoring updates your diagram as well as your source code.If you update your code in Eclipse, your diagram is updated as well there is no need to reverse engineer source code.Your source code and libraries are the model that is displayed, they are not reverse engineered into a different format.Several unique features make this possible: 
That is as accurate and up-to-date as your text editor, while being The UML notation to show a graphical representation of existing code The ObjectAid UML Explorer is different from other UML tools. It is easily installed from the repository: Name: ObjectAid UML Explorer  |
 RSS Feed
RSS Feed
